Reaction Thermodynamics calculates property changes in a reaction, such as ΔG, ΔH and ΔS, and equilibrium constants or electrode potential if electrons involve in the reaction. It can also calculate thermodynamics properties of individual chemicals. The function of Cp uses 7-parameter format:
Cp/R= a1T-2 + a2T-1 + a3 + a4T + a5T2 + a6T3 + a7T4
H and S are calculated using the following equations:
H/(RT)= -a1T-2 + a2T-1ln(T) + a3 + (1/2)a4T + (1/3)a5T2 + (1/4)a6T3 + (1/5)a7T4 + b1/T
S/R= -(1/2)a1T-2 - a2T-1 + a3ln(T) + a4T + (1/2)a5T2 + (1/3)a6T3 + (1/4)a7T4 + b2
Chemical Equilibrium uses Gibbs Free Energy Minimization method to calculate equilibrium compositions. Mathematically, Gibbs free energy minimization method can be expressed as:
(i=1, 2 ......N)
(k=1, 2 ......w)
The current version can only compute ideal gas equilibrium compositions. More advanced capability for multi-phases/non-ideal solutions will be in the future development.
Water Properties use Industrial Formulation 1997 of The International Association for the Properties of Water and Steam (IAPWS) to calculate thermodynamics properties of water and steam, such as H, U, S, Cp and speed of sound. IAPWS divides water properties into five regions as shown below.
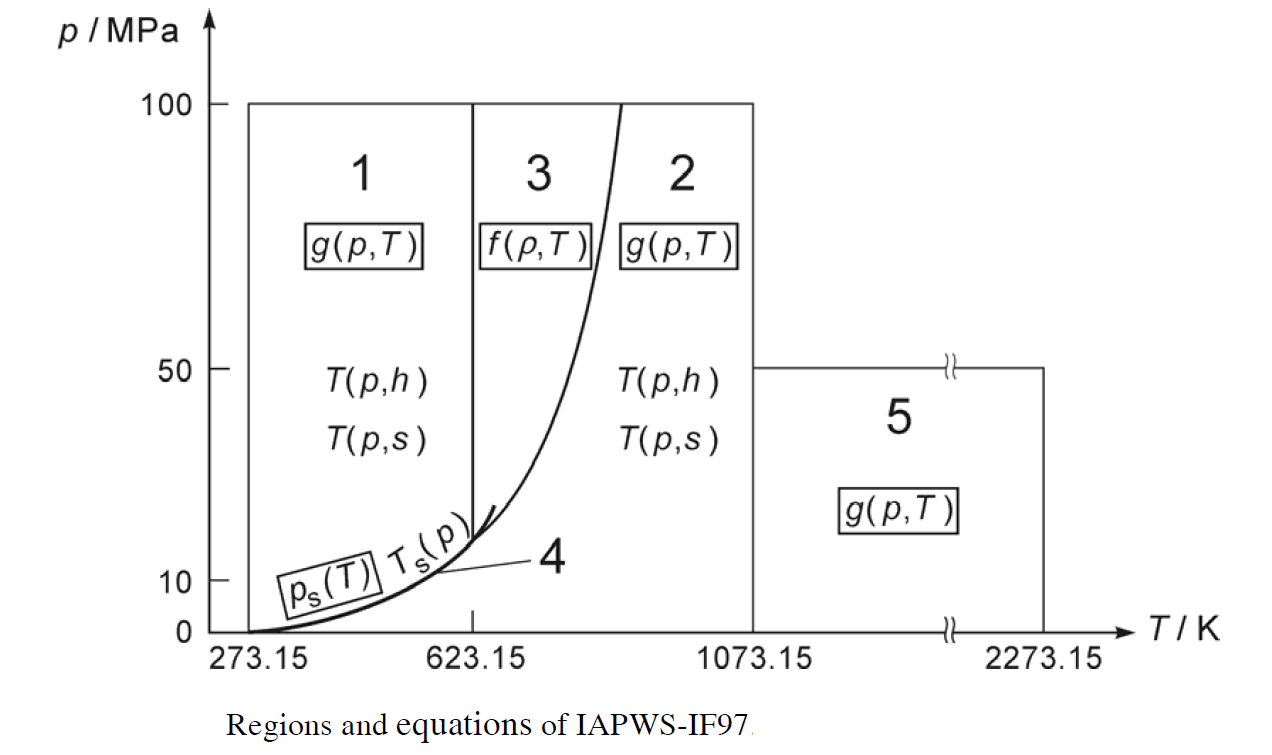
Each region has a set of complicated equations for the estimation of thermodynamics properties of water. If you are interested in more details, you can download the formulation. At the present, only region 4 for saturated steam is implemented. Saturated water of region 4 and all other regions are in the plan to be implemented.
Thermobook.net provides the following convenient tools:
Formula Weight Calculator
Chemical Equation Balancer
pH Calculator
Formulae with element coefficients in decimals and fractions are quite common in scientific papers and engineering formulations. The Formula Weight Calculator can handle all situations: integers,fractions and decimals.
Like the Formula Weight Calculator, the Chemical Equation Balancer can balance equations with element coefficients in integers, decimals and fractions. Between fractions and decimals, try to use fractions first if they are available because fractions are lossless.
The pH calculator provides convenient ways to calculate pH. Dissociation constants in the form of pKa for common acids and bases are also available by a simple click. You can also use your own dissociation constants. The input format of the dissociation constants can be numbers or strings in format like: 9.8x10^-5
A) Conventions for Formula Weight Calculation and Equation Balancer
In general, you just need to type in a formula and then go. However, when a formula is a composite compound, following
conventions are used:
1) Using * to formulate composite compounds such as CuSO4*0.5H2O. Dot (.) is reserved as decimal point.
2) A number at the beginning of a formula is interpreted as multiple of the whole formula, eg: 2CuSO4*0.5H2O = (CuSO4)2*H2O.
To make the multiple (2 in this case) just for CuSO4, you can write the formula as (CuSO4)2*0.5H2O.
3) Both fractions and decimals are acceptable. Both CuSO4*1/2H2O and CuSO4*0.5H2O will produce the same results.
B) Conventions for Thermodynamics Calculations
Thermodynamics calculations (Reaction Thermodynamics and Equilibrium Composition)
require complete information, such as state, charge, phase etc., for each chemical.
The following conventions are used for thermodynamics calculations:
1) Enclose charges and states in order in curly brackets { } immediately after a formula. if a chemical
is neutral, only state is needed.
2) Use the following rules to represent states:
l --- liquid
g --- gas
aq --- aqueous solution
s,cr,a,b,c,alfa,beta,... --- solid
3) Use e{-} to represent electrons. State may also be included, such as e{-g}.
4) Use * to formulate composite compounds, for example: CuSO4*3H2O, CuSO4*7/2H2O, CuSO4*0.5H2O. Dot (.) is reserved for decimal numbers.
5) Integers, decimals and fractions can all be used in equations. Between decimals and fractions, lossless fractions should be the first choice.
Thermobook.net was established in December 2011 by Dr. Chunzhi He. Dr. He himself is a Sr. software developer proficient in the areas of Java, PHP, systems, web UI and mobiles. Dr. He earned his Ph.D. in Chemical Engineering from the University of Connecticut. He has been leading the development of this site and the mobile releases since its establishment.
If you have any questions/suggestions or you noticed any bugs, you can contact us by email at info@thermobook.net
Express Board
January 19, 2025:
Thermobook.net officially releases
Stoichiometry Pro 1.0.
July 28, 2019:
Linux virtual boxes built from scratch using instructions from Linux from Scratch 8.4 and Beyond Linux from Scratch 8.4 are available to
download
February 24, 2019:
Thermobook.net starts Big Data Series 2: Globe Events. The project headlines globe events collected by GDELT project. Updates occur in 15 min interval
December 30, 2018:
Thermobook.net starts Big Data Series 1: Data Live with Spark. The project live streams AWS public datasets/XETRA dataset using Apache Spark along with WebSocket.
May 19, 2018:
Thermobook.net releases android application for stoichiometric calculations and more
Stoichiometry Plus
May 10, 2017:
Thermobook.net releases android application for molecular weight calculation: Thermo FW Calculator
April 30, 2017:
Thermobook.net releases
pH Calculator
April 8, 2017:
Thermobook.net completes migration of web host to AWS
April 29, 2014:
Thermobook.net launches
Solution Preparation Calculator
Nov 23, 2013:
Reaction Stoichiometry Calculator is officially launched